Abstract
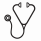
BACKGROUND: High-risk acute myeloid leukemia (HR-AML) is defined as AML with poor risk cytogenetics, antecedent hematologic disorders, and/or prior myelotoxic therapies. Median survival in HR-AML following standard cytarabine and anthracycline based intensive chemotherapy (IC) is approximately 6 months (JCO 2016,34:15_suppl,7000). Given the high incidence of HR-AML diagnoses in older AML patients (pts) with comorbidities and lower performance status, hypomethylating agents (HMA) are often used as an alternative induction regimen. However, to date, it is largely unknown how effective HMA induction is as compared with standard IC in this subset of pts.
OBJECTIVES: To retrospectively review the outcomes of HMA vs IC induction in older pts with HR-AML.
METHODS: We conducted a retrospective record review of consecutive pts ≥ 60 yrs old with newly diagnosed HR-AML treated with cytarabine/anthracycline based IC or HMA (azacitidine, decitabine) as upfront therapy at our institute. HR-AML was defined as: following prior chemotherapy for any reason (t-AML), history of prior myeloid malignancy such as MDS/MPN (s-AML), or characterized by poor risk cytogenetics indicative of AML with myelodysplasia-related changes (AML-MRC). Data was collected between January 2008 and December 2016. Demographics, initial hematologic parameters, cytogenetics, comorbidities, treatment response, subsequent allogeneic stem cell transplant (alloSCT), and overall survival (OS) were compared by induction treatment regimens.
RESULTS: We identified 201 HR-AML pts (98 treated with IC, 103 with HMA) at our institute (Table 1). 35 pts received azacitidine and 68 decitabine. IC-treated pts were younger (median age 68 yrs, range 60-85 yrs) vs HMA-treated pts (median age 74 yrs, range 63-93 yrs) with lower incidences of AML-MRC (IC 62% vs. HMA 73%) and t-AML (IC 27% vs. HMA 45%), but higher rates of s-AML (IC 48% vs. HMA 31%). Higher percentages of HMA-treated pts had complex karyotype (HMA 58% vs IC 49%) and monosomal karyotype (HMA 48% vs. IC 24%).
Pts with low Charlson Comorbidity Index (CCI) score (0 or 1) were 1.5 times more likely to get IC than HMA. HR-AML pts achieved higher complete remission (CR) rates following IC than HMA (IC 39% vs. HMA 27 %; p=0.10) although this was not significant. Substantially more pts receiving IC subsequently underwent alloSCT (IC 33% vs. HMA 5%). CR did not differ following IC vs HMA in pts stratified by age groups (60-64, 65-69, 70-95 yrs) or by AML-MRC, s-AML, or t-AML. After adjusting for alloSCT as a time dependent covariable, OS was not significantly better following IC than HMA induction (HR=0.75, 95% CI: 0.55 to 1.02; Table 2). Additional adjustments for ECOG performance status, CCI score, and age similarly showed no OS advantage in favor of IC over HMA in the entire study population (HR=0.84, 95% CI: 0.60 to 1.16) or in the various age groups analyzed. OS advantage favoring IC was seen in pts with t-AML (HR=0.56, 95% CI: 0.33 to 0.97) . This difference was maintained even with additional adjustments for ECOG, CCI, and age (HR=0.46, 95% CI: 0.28 to 0.87). In pts with presence of both complex and monosomal karyotypes (CK+MK+), OS advantage favoring HMA over IC was seen (HR=2.00, 95% CI: 1.08 to 3.70) which was maintained even with additional adjustments for ECOG, CCI, and age (HR=2.57, 95% CI: 1.26 to 5.22).
CONCLUSIONS: Our results suggest that induction therapy with IC is not superior to HMA for the majority of pts ≥ 60 yrs old with HR-AML. These results were not affected after adjusting for pt age, performance status, or comorbidity indices. The only possible exception were older pts with t-AML who preferentially benefited from IC. For all other older HR-AML pts, HMA may represent a feasible alternative upfront regimen with equivalent clinical outcomes, especially for transplant-ineligible individuals. Of interest, HMA therapy had superior survival advantage in pts with CK+MK+ HR-AML. These results should be validated in larger, multi-center prospective clinical trials. The impact of molecular profiling on these results will need to be further ascertained.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal